Photocatalysis is a fantastic way to clean facilities,
houses, and living environments. By modifying and further developing
this technology, we can reduce pollution in our air and water.
We can even reduce the spread of infections and diseases such
as SARS in hospitals. This cleaner way of life would benefit everyone
around the world.
The word photocatalysis is composed of two parts:
-
The prefix photo, defined as "light",
-
Catalysis is the process where a substance participates in
modifying the rate of a chemical transformation of the reactants without
being altered in the end. This substance is known as the catalyst
which increases the rate of a reaction by reducing the activation energy.
Hence, photocatalysis is a reaction which uses light
to activate a substance which modifies the rate of a chemical reaction
without being involved itself.
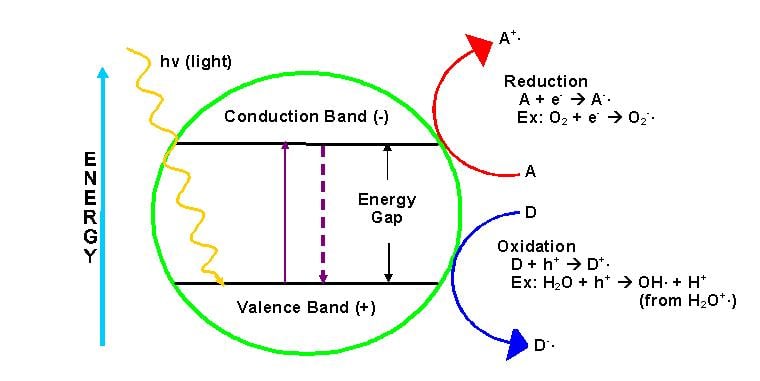
Today, semiconductors are usually selected as
photocatalysts, because semiconductors have a narrow gap between the valence
and conduction bands. In order for photocatalysis to proceed, the
semiconductors need to absorb energy equal to or more than its energy gap. This
movement of electrons forms e-/h+ or negatively
charged electron/positively charged hole pairs. The hole can oxidize donor
molecules.
Among the possible semiconductors, TiO2, or
Titanium Dioxide, (Eg
= 3.2 eV) is most extensively used because it has many advantages. It
is inert and resistant to corrosion, and it requires little post-processing,
making it inexpensive. Finally, it can react under mild-operating
conditions. However, it currently needs to use ultraviolet light for
photocatalysis to occur.

