







|
|
In order to return functionality to the infarcted myocardium, scientists
and doctors have recently been trying to take advantage of the current
revolutions in stem cell research. Scientists have attempted to
transplate a number of stem cell types into the damaged area of the
heart in order to promote regenesis, but the results thus far have been
dismal:
- A metastudy published in Nature reveals the
145 stem cell transplant attempts in animal models have been
disappointing failures
- Research demonstrates that the area of
implantation is so ischemic (lacking in oxygen and nutrients) that
less than 10% of stem cells remain after 7 days following
implantation
- Moreover, even these stem cells are so
stressed that their ability to differentiate into myocardiocytes
(heart muscle cells) is significantly impaired
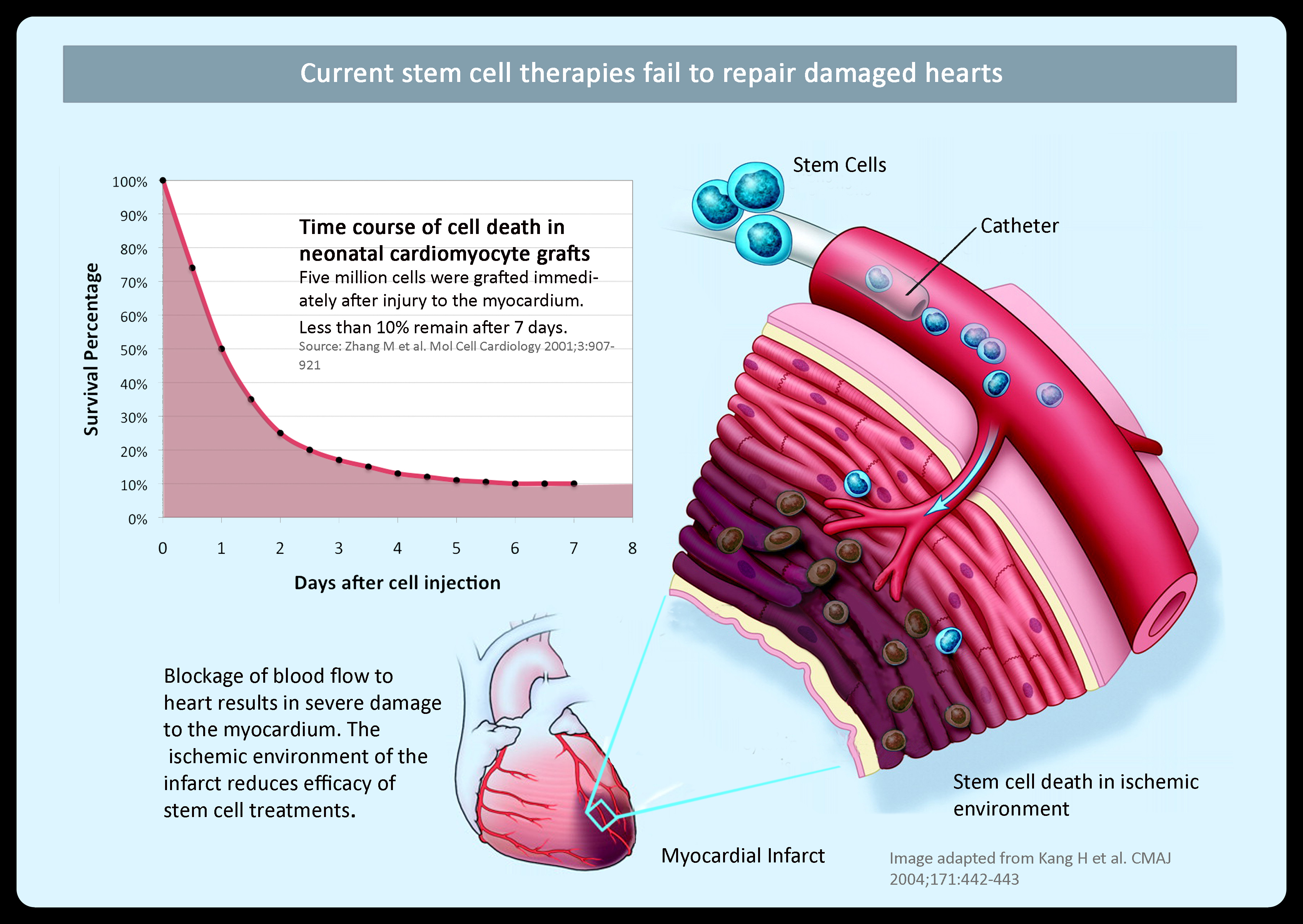
Clearly, despite its significant potential, the
efficacy stem cell therapy largely depends on relieving the ischemic
conditions of the damaged myocardium. Our technology aims to address
this challenge by taking advantage of a protein called vascular
endothelial growth factor (VEGF). VEGF promotes a process called
angiogenesis, or the growth of new blood vessels that can replenish
nutrients and oxygen to the damaged areas. However, systemic
administration of VEGF can be dangerous because of the high risk for
nonspecific, adverse side effects. Our technology overcomes this
challenge by delivering VEGF directly to the myocardium.
But before we move on, let's explore the history
of heart disease and MI as well as the evolution of our ability to
diagnose, prevent, and treat these conditions...


|


![]()