Future Technology
SMAGALS, or
Stop Myelin-Associated Glycoprotein for Amyotrophic Lateral Sclerosis, is a
futuristic combination of drugs and a pump that will help to improve the quality of
life for ALS patients, hopefully, by 2023. Much of the current research for ALS
is still in the hypothetical stages and has not yet been tested on humans.
We merged ideas from several medical school research groups with a unique
molecular cleavage mechanism and a pump to create SMAGALS.
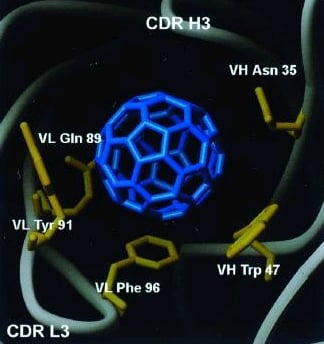 In
1985, the C60 buckminsterfullerene was discovered. See the blue
model of the buckyball on the left. Although in
the past, most applications of this molecule had been almost exclusively in
engineering, now
it is also used in biology. C60 spheres ("buckyballs")
have been modified by the addition of three axial, or equatorial, malonic acid
groups per molecule, and are a potentially powerful "free radical sponge." The
buckyball becomes water-soluble when it is chemically modified by the addition
of the acid groups. Research shows that the carboxyfullerenes are
neuroprotective in cell culture.
In
1985, the C60 buckminsterfullerene was discovered. See the blue
model of the buckyball on the left. Although in
the past, most applications of this molecule had been almost exclusively in
engineering, now
it is also used in biology. C60 spheres ("buckyballs")
have been modified by the addition of three axial, or equatorial, malonic acid
groups per molecule, and are a potentially powerful "free radical sponge." The
buckyball becomes water-soluble when it is chemically modified by the addition
of the acid groups. Research shows that the carboxyfullerenes are
neuroprotective in cell culture.
When tested in the transgenic model of familial
ALS, the carboxyfullerene with axially oriented malonic acid groups best delayed
the onset of disease and extended survival. The prolongation of survival rate
was about the same as that seen previously with Riluzole, which is thought to
act through the inhibition of excitatory neurotransmission. Thus, water-soluble
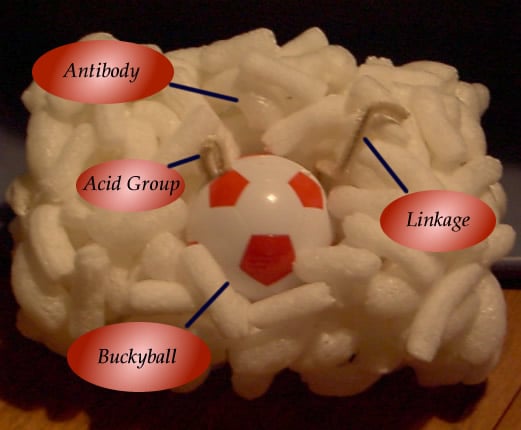 buckyballs
may prove to be useful neuroprotective agents. We created SMAGALS to use
the malonic groups attached to the buckyball to make it soluble in the spinal
fluid. The buckyball will deliver a new antibody for ALS therapy.
buckyballs
may prove to be useful neuroprotective agents. We created SMAGALS to use
the malonic groups attached to the buckyball to make it soluble in the spinal
fluid. The buckyball will deliver a new antibody for ALS therapy.
Since 2000, scientists have identified and
modeled the plausible binding site for a C60 fullerene to an
antibody. The massive antibody covers approximately 90% of the surface of the
buckyball, nearly burying the fullerene. Our prototype is displayed in the
picture on the right. Complete descriptions of the
exact geometry of the fullerene-antibody linkage have not been determined yet.
SMAGALS uses a link between the buckminsterfullerene and the antibody that is
sensitive to 600 nanometers (nm) of wavelength.
The bonding sites between the buckyball and
antibody contain very sensitive linkers that will break, due to energy transfer
(ET). This energy transfer will be sufficient to cause the bonds between
the buckyball and the antibody to break and free the antibody to stop the
myelin-associated glycoprotein (MAG) from working in the body.
An implantable, programmable, battery-powered
pump
will deliver the buckyballs into the spinal fluid.
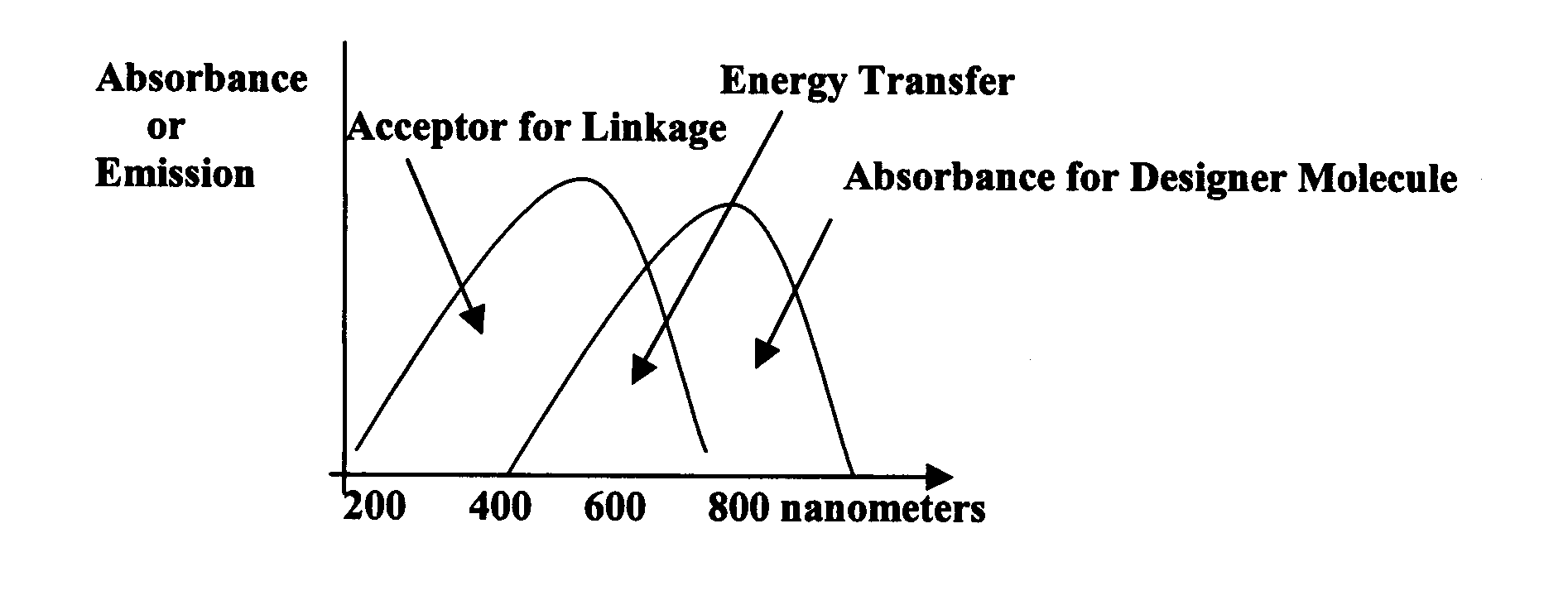 This pump will be placed
near the waist, in the back of the patient. A computer program will operate the
delivery of the drugs. Similar pumps are now used to help people with chronic
back pain. The buckyball pump (BP) will deliver C60 molecules, as it
is linked to an antibody as well as a designer molecule. The BP will be
programmed to deliver a designer molecule that has absorbed two or three 800 nm
photons from the IR diode as the molecules pass through the pump. ET will
occur at about 600 nm when the designer molecules are near the
buckyball-antibody linkage.
This pump will be placed
near the waist, in the back of the patient. A computer program will operate the
delivery of the drugs. Similar pumps are now used to help people with chronic
back pain. The buckyball pump (BP) will deliver C60 molecules, as it
is linked to an antibody as well as a designer molecule. The BP will be
programmed to deliver a designer molecule that has absorbed two or three 800 nm
photons from the IR diode as the molecules pass through the pump. ET will
occur at about 600 nm when the designer molecules are near the
buckyball-antibody linkage.
The new antibody for SMAGALS blocks
myelin-associated glycoprotein (MAG). Recent research has already identified
these glycoprotein molecules, which have inhibiting effects on nerve
regeneration. The SMAGALS antibody will block MAG, neutralizing these inhibitors
and helping nerve regeneration. Normally, the body controls the number of
contacts nerve cells make by not letting new connections be made. When the
spine is injured, the body needs to repair the nerve cells and its long axons,
and so the chemicals in the body that stops regeneration of nerves needs to be
shut down. MAG is one of the “stop signs” in the body; SMAGALS is designed to
make MAG ineffective. This allows the body to regenerate new myelin wrappings
around the nerve cells. Since ALS is a disease that gradually destroys myelin
cells, this method of stopping MAG will greatly benefit people who suffer from
ALS.
SMAGALS is a futuristic approach to promote a
better quality of life for patients with ALS. It is NOT a cure. Since the
cause of the disease is still unknown, one can only approach the problem by
interfering with one of the proposed mechanisms in the body. SMAGALS uses
up-to-date research and then projects these ideas into technologies that might
be feasible by the year 2023. Using the C60 molecule to trap free
radicals, as well as making it a vehicle to move the new antibody into the
spinal fluid is a potentially wonderful technology to help defeat ALS. The
buckyball pump delivers the photosensitive linkage between the buckyball and
antibody, and a designer molecule that absorbs IR photons. Inhibiting the work
of MAG by the new antibody allows the body to regenerate nerve cells. The motor
neurons will begin to function and let the person use his/her limbs.
Below is a Quicktime movie clip that shows the photon (laser light) as it breaks
the linkage between the buckyball and antibody:
 In
1985, the C60 buckminsterfullerene was discovered. See the blue
model of the buckyball on the left. Although in
the past, most applications of this molecule had been almost exclusively in
engineering, now
it is also used in biology. C60 spheres ("buckyballs")
have been modified by the addition of three axial, or equatorial, malonic acid
groups per molecule, and are a potentially powerful "free radical sponge." The
buckyball becomes water-soluble when it is chemically modified by the addition
of the acid groups. Research shows that the carboxyfullerenes are
neuroprotective in cell culture.
In
1985, the C60 buckminsterfullerene was discovered. See the blue
model of the buckyball on the left. Although in
the past, most applications of this molecule had been almost exclusively in
engineering, now
it is also used in biology. C60 spheres ("buckyballs")
have been modified by the addition of three axial, or equatorial, malonic acid
groups per molecule, and are a potentially powerful "free radical sponge." The
buckyball becomes water-soluble when it is chemically modified by the addition
of the acid groups. Research shows that the carboxyfullerenes are
neuroprotective in cell culture. 
